Focus on Error Prevention: Identical Drug Identification Numbers
By Ian Stewart B.Sc.Phm., R.Ph.
Pharmacy professionals usually use the assigned Drug Identification Number (DIN) of a drug product to confirm that the correct product is being dispensed.
The DIN is a computer-generated number assigned by Health Canada to a drug product that uniquely identifies the following product characteristics: manufacturer; product name; active ingredient(s); strength(s) of active ingredient(s); pharmaceutical form; and route
of administration1.
Pharmacists should be aware of the potential for error when two different product formulations are assigned the same DIN.
Case:
Rx:
Amoxicillin 125mg/5ml
Sig: Give 9mls three times daily
Mitte: 10 days’ supply
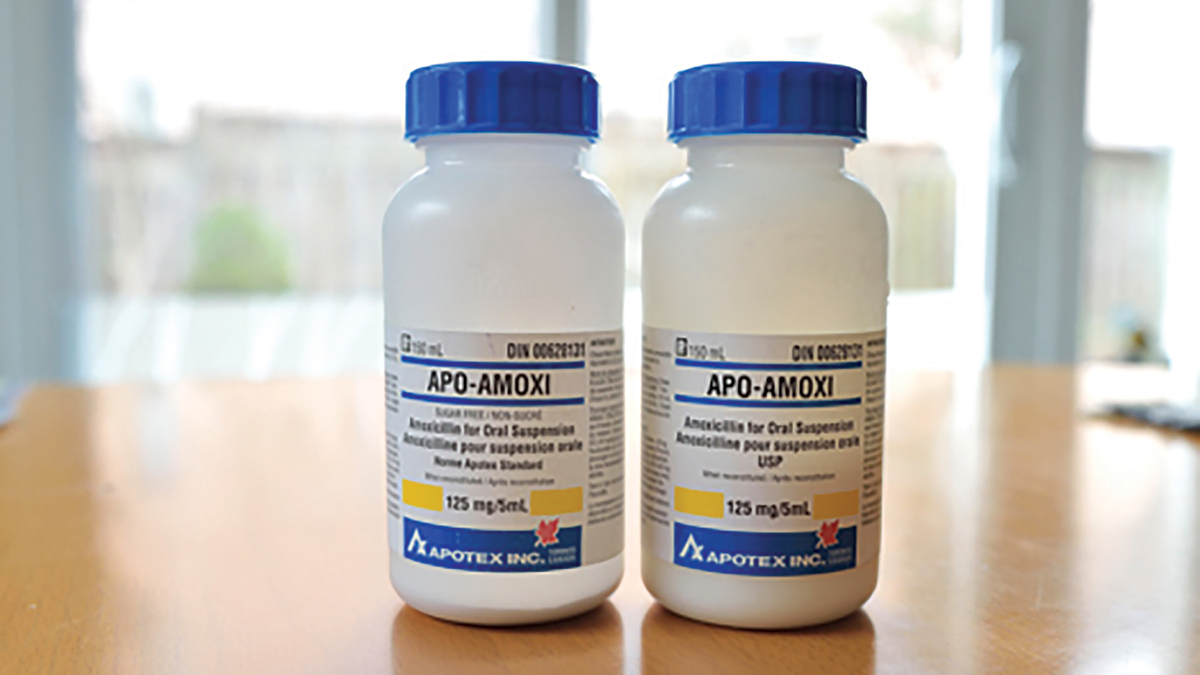
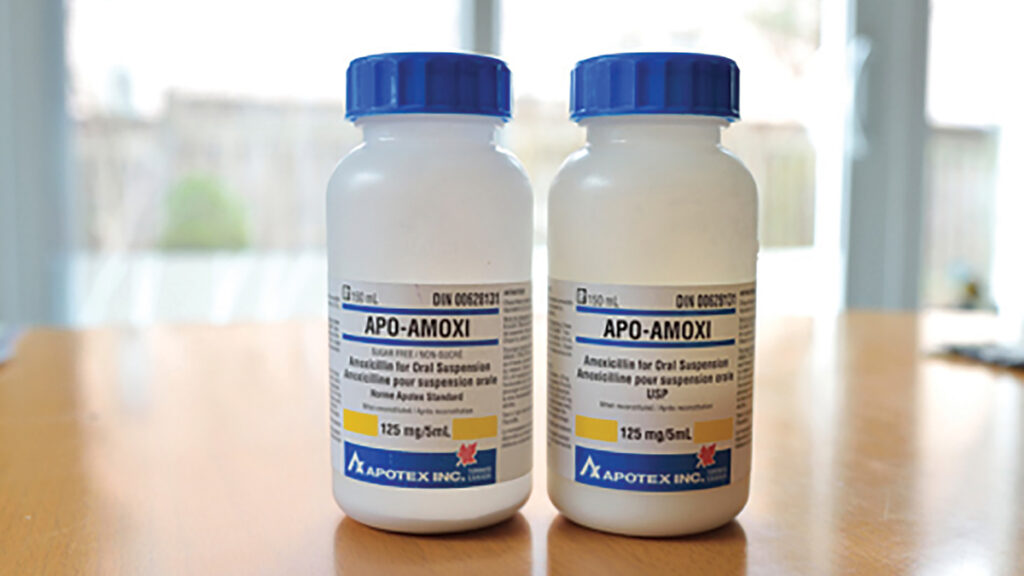
The above prescription was taken to a local community pharmacy for dispensing. A pharmacy assistant correctly entered the prescription into the computer. However, when preparing the medication for dispensing, the pharmacy assistant selected one 150ml bottle of Apo-Amoxi®125mg/5ml and one 150ml bottle of Apo-Amoxi®125mg/5ml sugar free in error. Both products are similar in appearance and have the same DIN (see photograph).
To reconstitute the 150ml bottle of Apo-Amoxi®125mg/5ml, the pharmacy assistant added 103mls of water as per the label instructions. The pharmacy assistant added the same volume of water to the 150ml bottle of Apo-Amoxi®125mg/5ml sugar free. However, the final volume was much less than 150mls.
Upon checking the label of the sugar free product, it was noted that 148mls of water (not 103mls) should be added to reconstitute the product. The error was therefore detected.
Possible Contributing factors:
- Pharmaceutical manufacturers often use similar packaging and labelling for consistency. As a result, the packaging and labelling of Apo-Amoxi®125mg/5ml and Apo-Amoxi®125mg/5ml sugar free are similar in size, shape and appearance.
- Both products have
identical DINs. - The ‘sugar free’ indication on the label of the Apo-Amoxi®125mg/5ml sugar free is not bolded and therefore easy to miss.
- Both products were stored together in the same location.
- The pharmacy assistant assumed that both products were identical and hence assumed the instructions for reconstitution would be the same.
Recommendations:
- Educate all pharmacy staff about the potential for error when storing and dispensing drugs with identical DINs.
- Store the sugar free product in a separate location which should be clearly labelled.
- Consider changing the brand of one of these products to reduce the potential for error.
- Due to the similarity in a manufacturer’s packaging and labelling, when dispensing multiple products, check each carefully to ensure they are all the same product.
- Consider having the pharmacist or technician double check the products selected and/or the amount of water to be added prior to reconstitution.
- Check the prescription for accuracy and appropriateness before reconstituting the product.
- Implement scanning technology to verify products by UPC, which is unique to each formulation.
References:
- Drug Identification Number (DIN), available at: https://www.canada.ca/en/health-canada/services/drugs-health-products/drug-products/fact-sheets/drug-identification-number.html
Accessed June 30th, 2017.
Please continue to send reports of medication errors in confidence to Ian Stewart at: [email protected]. Please ensure that all identifying information (e.g. patient name, pharmacy name, healthcare provider name, etc.) are removed before submitting.
Note: Presently, Apotex has two approved DINs listed for Apo-Amoxi in Health Canada’s Drug Product Database. Apotex has confirmed that only one of the products is available for sale (DIN 00628131).