Non-Sterile Compounding
Yes, this course is free of charge and open to anyone. Anyone who wishes to supplement or refresh their training is encouraged to take the course (e.g., moving to a new pharmacy with a different level of requirements in place, returning to the non-sterile compounding supervisor role after an extended leave).
Registrants who choose to complete this training course voluntarily are not required to self-declare in the OCP Portal and can retain their certificate of completion for their own records.
New non-sterile compounding supervisors will need to sign in to the OCP Portal and confirm they have completed the mandatory training under the Authorized Training tab.
The final exam must be taken to generate a certificate of completion. Non-sterile compounding supervisors should retain a copy of their certificate for their records and ensure it is available to the College upon request, for auditing purposes. The College will collect the documentation it needs to verify the requirements have been met when it is appropriate to do so.
College operations advisors will follow-up with non-sterile compounding supervisors whose pharmacies have not met the standards after their pharmacy assessments to confirm completion of the training course.
New compounding supervisors will need to self-declare they have completed the mandatory training course. (See FAQ: “I am starting a new role as a compounding supervisor. How do I self-declare that I have completed the training?”)
New non-sterile compounding supervisors are required to complete the training course within three months of starting the role, although it is preferred that they complete the training course before starting.
Current non-sterile compounding supervisors whose pharmacies are not meeting the standards will have one month to complete the training course; however, College operations advisors may extend this period as appropriate to the circumstances.
The final exam must be taken in order to confirm completion of this mandatory training course.
The mandatory training goes into effect immediately: February 5, 2026.
Current non-sterile compounding supervisors in community and hospital pharmacies are required to complete the training course to address identified gaps if an operational assessment reveals that a pharmacy does not fully meet the standards. In this event, the College operations advisor will assign the training course as part of the action plan in the pharmacy’s assessment report.
The non-sterile compounding supervisor training course was designed to be taken in its entirety; however, there may be circumstances in which College operations advisors recommend or require completion of select modules from the training course. These will be handled on a case-by-case basis.
A “new” non-sterile compounding supervisor is defined as someone who is new to the role. Any pharmacist or pharmacy technician who is assigned as a non-sterile compounding supervisor on or after February 5, 2026 is responsible for taking this mandatory training course. If you became a new compounding supervisor before this date, this requirement does not apply. However, we strongly encourage all registrants in a non-sterile compounding supervisor role take the training course to meet any self-identified learning needs and goals (e.g., if you feel like you need to reinforce your existing training, are coming back from an extended leave, or are taking this position in a new pharmacy with a higher level of requirements in place).
In a community pharmacy, the Designated Manager (DM) is the non-sterile compounding supervisor unless they have assigned this role to another pharmacist or a pharmacy technician.
In a hospital setting, the pharmacy manager/department head of the hospital pharmacy is the non-sterile compounding supervisor, unless they have assigned this role to a pharmacist or pharmacy technician.
At the March 2024 Board meeting, the Board approved mandatory training for new compounding supervisors in all pharmacies and current compounding supervisors in pharmacies where standards are not being met. The decision to require OCP-approved training was in response to operational assessment data showing standards are not being fully met, in part due to insufficient training of compounding supervisors. This decision follows the principles of right-touch regulation in which a regulatory response is required if the risk of harm to patients is high.
Health Canada considers compounding to be the following:
“The combining or mixing together of two or more ingredients (of which at least one is a drug or pharmacologically active component) to create a final product in an appropriate form for dosing. It can involve raw materials or the alteration of the form and strength of commercially available products. It can include reformulation to allow for a novel drug delivery. Compounding does not include mixing, reconstituting, or any other manipulation that is performed in accordance with the directions for use on an approved drug’s labelling material.”
Before compounding a non-sterile preparation, the need for the compounded product should be confirmed by checking for commercially available preparations in the Health Canada’s Drug Product Database and contacting manufacturers. To comply with the Health Canada policy on compounding, this confirmation is required in order to validate the lack of product availability and avoid duplicating an approved drug.
Non-sterile preparations can be categorized as simple, moderate or complex (as outlined in United States Pharmacopeia (USP) Chapter <795> Pharmaceutical Compounding – Nonsterile Preparations). A number of factors go into determining the type of preparation and level of risk when compounding preparations. Pharmacists and pharmacy technicians who compound non-sterile preparations should evaluate their practice, develop service-related procedures and implement appropriate quality controls for both patients and compounding personnel, with a view to guaranteeing the overall quality and safety of non-sterile preparations.
The National Institute for Occupational Safety and Health (NIOSH) website explains that a hierarchy of controls is way to identify a preferred order of actions to best control hazardous workplace exposures. Compounding supervisors should review the descriptions of each level of control. The diagram below* provides examples of what the various controls might be feasible in the context of pharmacy compounding to minimize risk of exposure to occupational hazards.
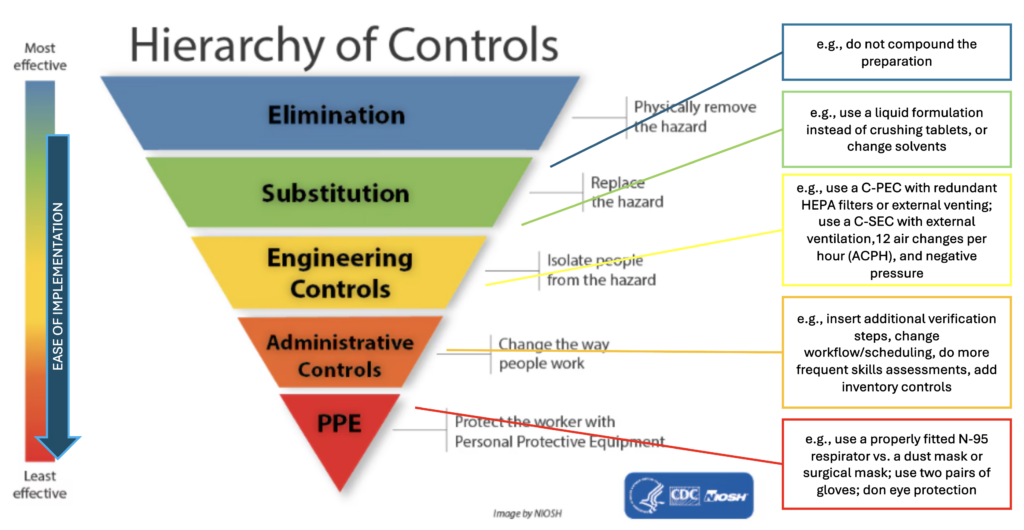
*Adapted from: https://www.cdc.gov/niosh/topics/hierarchy/default.html.
It is the responsibility of the compounding supervisor and manager to evaluate and choose the appropriate controls, depending on the type of risk posed by a hazardous product. Key points to consider in your decision-making are:
- The level of risk the ingredient(s) may present
- The volume/frequency of each ingredient used
- The combined exposure to all higher risk ingredients
Each prescription and patient situation must be assessed individually. In the event it is necessary to compound a particular preparation requiring processes and safety measures that are not currently in place in the pharmacy, the documentation should specify the following:
- Potential risks of compounding the preparation without the processes and safety measures currently in place
- The extra steps that must be taken to mitigate these risks
- References confirming that these steps will actually minimize risks to the quality of the product and safety of personnel
Procedures for mitigating risk must be documented in the master formulation record and reviewed at least every 12 months.
Refer to sections 9, 9.1.1, 9.2.1, 9.2.3 and 9.6 of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations and the Pharmacy Connection article — A Closer Look at Personal Protective Equipment. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.
The NAPRA Non-Sterile Compounding Standards do not assign a specific numerical value to define “small quantity” or “occasional.”
What constitutes an “occasional small quantity” for a preparation or ingredient depends on the amount handled and how frequently it is used; however, these cannot be considered in isolation.
First, amounts and frequency are not the only factors involved in assessing the overall degree of risk to pharmacy staff and patients (and subsequently the appropriate level of requirements [A, B or C] for a preparation that contains a hazardous product).
Second, the cumulative risk of exposure over time must also be considered, even if the preparations are compounded on different days. The level of requirements must be sufficient for the pharmacy’s compounding practice as a whole to minimize contamination of compounded products and to provide adequate protection for compounding personnel, other staff and patients entering the premises.
For example, if a pharmacy compounds many different products “occasionally,” it is possible for their level of requirements to be the same as that of a pharmacy that compounds a few products “routinely.” When it doubt, err on the side of caution. If there is uncertainty about which risk level to assign, go with the higher standard – quality patient care and safety for all personnel should always be the top priority.
The Designated Manager (DM), pharmacy department head or non-sterile compounding supervisor (if one has been appointed) is responsible for determining whether an amount is “small” or “large,” or a frequency is “occasional” or “routine.” Conducting a risk assessment for each compounded preparation will help you determine the most appropriate level of risk, while documenting these risk assessments can help you support your rationale during a pharmacy operational assessment.
Refer to Section 4.1 and 4.2, Section 9 of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.
Schedule 2 of the Hazardous Products Act (HPA) lists the health hazard classes, not individual products. The health hazard(s) for an active pharmaceutical ingredient (API) are identified on its safety data sheet under Section 2 – Hazards Identification.
The health hazard classes can be identified by a pictogram (see an example below), which also appears on the API’s label.

Safety data sheets provide information about a product’s risks and include recommended preventive measures to protect staff, such as engineering controls and personal protective equipment (Section 8). This is helpful in identifying products that are volatile, aromatic, sensitizing or very irritating to the respiratory tract, skin or mucous membranes as part of the risk assessment and determining the level of requirements needed.
The information in the safety data sheet must be interpreted and applied in the context of the pharmacy’s compounding practice. Compounding supervisors must consider the potential exposure of both staff and patients to the product (e.g., the physical and characteristics and chemical properties of the API, the frequency and duration of manipulation, and the potential for contact).
Note: Safety data sheets are available from the API supplier or through the Canadian Centre for Occupational Health and Safety and are updated every three years. They should be readily accessible and retrievable, and kept in a location known to all pharmacy personnel.
Manufactured drugs approved by Health Canada (i.e., those with a drug identification number [DIN]) are exempt from the HPA and therefore do not have safety data sheets. If a Health Canada–approved drug is used in a preparation, the safety data sheet for the API(s) in that drug should be consulted. The drug’s product monograph should also be considered, as it includes information on storage, stability and disposal and special handling instructions
If a compound is made up of more than one API or drug, then all of the available safety data sheets and product monographs (if applicable) must be consulted to complete your risk assessment.
Refer to Section 4.3, 6.3.4, 8, 8.1, 8.2, 8.3 and Glossary of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.
The National Institute for Occupational Safety and Health (NIOSH) is a recognized occupational health and safety agency, and part of the U.S. Centers for Disease Control and Prevention. The NIOSH List of Hazardous Drugs in Healthcare Settings, 2024 places hazardous drugs into tables based on specific criteria.
Any drug or active pharmaceutical ingredient (API) that is not included on the NIOSH list did not meet their specific criteria for a hazardous drug. However, it is important to read the NIOSH document in its entirety (i.e., do not only refer to the list of drugs in each table). The actual risk to personnel depends on how the drug is handled and what risk mitigation measures are in place.*
The Hazardous Products Act (HPA) is the legislation that that enforces safety measures. It establishes hazard classifications and mandates that any supplier who sells or imports a hazardous product intended for use, handling or storage must provide cautionary labelling requirements and a safety data sheet (SDS).
The Workplace Hazardous Materials Information System (WHMIS) is Canada’s hazard communication standard to support the safe use of hazardous materials in the workplace. It includes products used in various workplaces – a far wider group of chemicals than NIOSH. These are categorized as either a physical hazard or a health hazard (see Schedule 2 of the HPA). It also identifies products that can be very irritating to the respiratory tract, skin or mucous membranes, which is important for determining the level of requirements.
Both NIOSH and WHMIS provide information on toxicities of drugs. The WHMIS also includes information on other workplace products. Both references should be consulted in the risk assessment process, as described in Section 4 of the guidance document and illustrated in the decision algorithm.
Safety data sheets and other applicable references should be reviewed, accessible and easily retrievable for every single ingredient in a compounded preparation (see next question).
Risk assessments should be done for each compounded preparation. They should be made available to all staff so they can review them and understand the risks.
*The NIOSH List is designed to help employers to identify which drugs handled by employees are considered by NIOSH to be hazardous drugs. Because new drugs and new formulations are continuously brought to market between NIOSH’s periodic updates, hazardous drug evaluation should be a continual process. Employers should establish their own procedures to identify and evaluate new drugs as they enter their workplace and, when appropriate, reassess their presence on hazardous drug lists as toxicological data become available to support re-categorization. (Source)
Refer to Sections 4.3 and 8.3 of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.
Ventilation (the “V” in HVAC) is an engineering control intended to remove or control contaminants released in indoor work environments by bringing in fresh air. The Canadian Centre for Occupational Health and Safety (CCOHS) Fact Sheet on Industrial Ventilation may be helpful for background information on this subject, such as the purpose of a ventilation system.
For non-sterile compounding, the Designated Manager, pharmacy department head or non-sterile compounding supervisor (if one has been appointed) must determine what ventilation is required for their pharmacy practice; they can do so through their risk assessment, taking into account cumulative risk.
Ventilation must be controlled in such a way as to avoid decomposition and contamination of chemicals while maintaining the quality and efficacy of stored products and ensuring the safety and comfort of compounding personnel. For example:
- Air vents should not be located directly over work areas, to avoid contamination of the products.
- Fans are unsuitable for ventilation as they merely blow the contaminant around the work area without effectively removing or controlling them.
- Opening a window or door might introduce outdoor air, but it is uncontrollable and risks bringing in additional contaminants and disrupting the compounding environment.
The external ventilation requirements differ between Levels A, B and C and are summarized below. Refer to the guidance document additional details. Note that “external” means the air must go outside the building (not outside the room).
| Level | External Ventilation | Guidance Document Section |
|---|---|---|
| All levels | The OCP’s Standards of Operation for Pharmacies require pharmacies to be designed, constructed and maintained to ensure the integrity and the safe and appropriate storage of all drugs and medications. This includes proper conditions of sanitation, temperature, light, humidity, ventilation, segregation and security. | n/a |
| A | External ventilation is not required for the designated compounding area Refer to the requirements for all levels of compounding | 5.4.1.3 |
| B | External ventilation is not required for the compounding room. However, this dedicated room must be entirely closed off (i.e., separate from the rest of the pharmacy) and well-ventilated If a ventilated containment device (C-PEC*) is installed in the room, the C-PEC should either be externally vented (the preferred option) or have redundant HEPA filters in series. | 8.2 9.1 9.2.1 |
| C | The room in which the C-PEC is placed (the C-SEC†) must be separate, under negative pressure, and have appropriate air exchange, with external ventilation through HEPA filtration. The C-PEC installed in the C-SEC should be externally vented (the preferred option) or have redundant HEPA filters in series. | 8.3 9.1.1 9.1.2 9.1.5 9.2.1 |
Compounding personnel should also understand how the secondary ventilation system operates (i.e., the HVAC of the C-PEC) and be able to recognize and address any problems with it.
*C-PEC – containment primary engineering control, designed to minimize the exposure of workers and the environment to hazardous products when such products are being handled directly.
†C-SEC – containment secondary engineering control (i.e., room in which the C-PEC is placed).
Refer to the Sections of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations referenced in the table below. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.
For hazardous non-sterile compounding, the level of requirements needed (i.e., B or C) depends more on the risk(s) posed by the hazardous product than on the complexity of the preparation.
In addition to the requirements for non-sterile compounding, such as adequate space and sufficient lighting (guidance document, section 5.4), hazardous non-sterile preparations should be compounded in a separate, dedicated room. For example, compounding hazardous products containing carcinogenic drugs listed in NIOSH† Table 1, or those categorized by WHMIS‡ as a health hazard because they are very irritating to the respiratory tract, skin and/or mucous membranes, should take place in a Level C room. This means a physically separate, closed-off room with external venting through HEPA filtration, appropriate air exchange and negative pressure to avoid contaminating the environment and to further protect personnel. There should also be an appropriate containment device (C-PEC) for materials being compounded.
Because of the difficulty of removing hazardous product contamination, the surfaces of ceilings, walls, floors, fixtures, shelving, counters and cabinets in the non-sterile compounding area should be smooth, impermeable, free from cracks and crevices and made of non-shedding material.
If a separate area is not possible and non-hazardous non-sterile preparations must be compounded in the same room as hazardous non-sterile preparations, at an absolute minimum there should be policies and procedures in place to ensure that the area is meticulously cleaned to prevention any risk of cross-contamination.
If you intend on compounding both non-hazardous and hazardous non-sterile preparations, refer to the FAQ “Do we need two separate designated compounding areas for hazardous and non-hazardous non-sterile preparations?” for additional considerations.
†National Institute for Occupational Safety and Health.
‡Workplace Hazardous Materials Information System.
Refer to Sections 9.1, 9.1.1, 9.2.1, 9.2.2 and 9.3 of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.
It is strongly recommended that the pharmacy has one set of dedicated equipment (including personal protective equipment) for compounding hazardous drugs and another set for non-hazardous drugs. Alternatively, disposable equipment should be used, if possible, to reduce the risk of cross-contamination.
If it is not possible to have dedicated equipment for hazardous compounding, at an absolute minimum there should be policies and procedures in place to ensure that equipment is meticulously deactivated, decontaminated and cleaned immediately after compounding preparations with hazardous materials, before being used for non-hazardous preparations. The pharmacy should review section 9 of the guidance document, which further outlines the requirements for hazardous preparations.
For occasional non-sterile hazardous compounding, a C-PEC* used for sterile hazardous compounding (e.g., Class II BSC or CACI†) may be used, provided it is decontaminated, cleaned and disinfected before compounding the non-sterile preparation and again before resuming sterile compounding in that C-PEC due to the higher risk posed to patients.
*C-PEC (containment primary engineering control): a ventilated device designed to minimize exposure of personnel and the environment to hazardous products when such products are being handled directly (guidance document, section 9.2). Refer to question below regarding facilities.
†BSC – biological safety cabinet; CACI – compounding aseptic containment isolator.
Refer to Sections 9.2.1, 9.2.2, 9.2.3 and 9.3 of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.
If there is uncertainty regarding the risk level to assign a compounded preparation after completing your risk assessment, defer to the higher standard in the interest of safety. The Designated Manager/non-sterile compounding supervisor’s rationale to support the pharmacy’s decision must be well-researched and documented in the risk assessment, with supporting references.
Refer to Section 4 of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.
No, the NAPRA Model Standards for Pharmacy Compounding of Non-Sterile Preparations (elsewhere referred to as “the standards”), adopted by the College, do not assign specific drugs, ingredients or formulations to each level of requirement (A, B or C). This is because compounding, like other aspects of pharmacy practice, is highly varied and dynamic.
For instance, risk assessments of similar formulations may yield different results across pharmacies because of the following:
- Numerous factors involved in assessing risk (e.g., quantity of ingredients being handled, frequency of compounding high-risk or low-risk preparations)
- Differences in pharmacy size, layout, personnel, number and volume of preparations compounded in the pharmacy, etc.
- Customized nature of compounded preparations, which may be altered to suit a specific patient’s needs
- Various ways risk may be mitigated (e.g., consider the NIOSH “hierarchy of controls”)
How a pharmacy chooses to conduct and document the rationale for their risk assessments so that it is reflective of their pharmacy is up to the discretion of the non-sterile compounding supervisor. For further support, the College has developed an optional risk assessment template, companion guide and examples for ensuring a risk assessment is performed for each compounded preparation.
Refer to Section 4.1 of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations for additional factors to consider that can have an impact on the outcome of your risk assessment. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.
UPDATED:The College has developed a risk assessment template, along with a companion guide and two examples (1% hydrocortisone powder in 1% clotrimazole cream and progesterone 400 mg suppositories) to help registrants understand what to document, reference, and consider when conducting a risk assessment. Use of the OCP template is optional, as how a pharmacy chooses to document risk assessments is at the discretion of the non-sterile compounding supervisor.
Every pharmacy that offers non-sterile compounding services must meet or exceed the minimum requirements set out in the NAPRA Model Standards for Pharmacy Compounding of Non-Sterile Preparations (elsewhere referred to as “the standards”).
The steps for conducting a risk assessment are described in Section 4 of the standards. In brief, there are three levels of requirements (A, B, and C), defined in section 8 (page 12) of the standards. These correlate to the compounded preparations’ associated risk and their complexity. The level of requirements the pharmacy needs to have in place depends on which non-sterile preparations are compounded, their complexity, how frequently they are compounded, at what concentration and in what amounts. Cumulative risk (of all compounded preparations) must be taken into account. Both risk to the preparation and risk to personnel must be considered and mitigated by adequate protection measures.
A risk assessment must be completed for each compounded preparation, taking into account every ingredient (e.g., drug, active pharmaceutical ingredient [API]) used.
A risk assessment should be conducted with appropriate resources. Safety data sheets and other applicable references should be consulted. The rationale for compounding and any risk mitigation strategies should be documented. Policies and procedures that ensure safety for personnel must be in place and documented as well.
If there is uncertainty on the risk level to assign, go with the higher standard. Quality patient care and safety of all personnel should always be the top priority.
Refer to Sections 4 and 8 of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.
Refer to Sections 9.1, 9.1.1, 9.2.1, 9.2.2 and 9.3 of the NAPRA Guidance Document for Pharmacy Compounding of Non-sterile Preparations.
It is preferable to have separate areas for compounding hazardous and non-hazardous non-sterile preparations, however, if this is impossible and the same area is used, compounding and/or cleaning personnel must, at minimum, be assured that the area and any reusable equipment has been meticulously deactivated, decontaminated and cleaned to prevent any risk of cross-contamination from the hazardous materials before other preparations are compounded.
Because of the difficulty of removing hazardous product contamination, the surfaces of ceilings, walls, floors, fixtures, shelving, counters and cabinets in the non-sterile compounding area should be smooth, impermeable, free from cracks and crevices, and made of non-shedding material. It is strongly recommended that equipment be dedicated for compounding each of hazardous and non-hazardous drugs. Alternatively, disposable equipment should be used, if possible, to reduce the chances of cross-contamination.
The Designated Manager/department head and/or non-sterile compounding supervisor must have policies and procedure in place for the deactivation, decontamination and cleaning required after compounding hazardous non-sterile preparations. As part of the pharmacy’s quality assurance program, personnel must be trained and their work routinely assessed to ensure compliance with procedures.
Refer to Sections 5.4.1, 9.1.1 and 9.2.1 of the NAPRA Guidance Document for Pharmacy Compounding of Non-sterile Preparations.
It is a standard of accreditation for all pharmacies to have two sinks (or one double sink) in the dispensary. Accredited pharmacies engaged in non-sterile compounding must also meet the NAPRA Non-Sterile Compounding Standards to have a clean water supply, with hot and cold running water, available in or close to the compounding area.
Please refer to the table below which summarizes the accompanying Guidance to meet the Standards.
Compounding must take place within the accredited pharmacy area. Any material changes to the existing accredited area or layout require a Notice of Renovation to be submitted to the College, and an Operations Advisor will be assigned to review the proposed floor plan.
| LEVEL | Sink Location | GD Section | Comments |
|---|---|---|---|
| A | In or close to the compounding area | 5.4.1.4 | If the designated compounding area is in the dispensary, the minimum sink requirement for accreditation may serve to meet both sets of standards. Consider accessibility (for example, compounding volume and number of personnel compared to the available time and space), ease of use, potential for splashing nearby areas, etc. |
| B | In the compounding room | 5.4.1.4 | If there is a C-PEC, consider Section 9.1.1 and consult supplier regarding proper installation and certification |
| C | In the compounding room Water sources and drains should be located at least 1 meter away from the C-PEC. | 5.4.1.4 9.11 | |
| All levels | Clean, potable, hot and cold running water for washing hands and equipment Preferably made of stainless steel and having touchless control | 5.4.1.4 | Plumbing system should be free of defects that could contribute to contamination |
Refer to Section 5.2, Table 1, Table 2, Checklist 1 and Table 8.4 of the NAPRA Guidance Document for Pharmacy Compounding of Non-sterile Preparations.
There is no requirement in the College-adopted NAPRA Non-Sterile Compounding Standards for personnel to complete a formal, accredited or third-party training program. The pharmacy manager/department head (or designated non-sterile compounding supervisor) may choose to develop their own training tools and assessment program to suit their specific needs. The intended outcome is that the expertise of personnel responsible for compounding must be commensurate with their assigned duties.
The potential need for training and routine assessment is not limited to the compounding processes or technique; personnel must also be educated on policies and procedures, such as those related to attire, personal protective equipment, cleaning, and conduct.
Refer to Table 8.4 of the Guidance Document for an overview of training topics for each Level (A, B or C), as well as the specific sections relevant to the type of personnel or preparation.
- Checklist 1: Skills assessment checklist for compounding process
- Table 1: Elements to cover in training of compounding personnel
- Table 2: Elements to cover in training of cleaning personnel
- Table 3: Examples of policies and procedures
For registrants interested in exploring external courses, the College provides a listing of continuing education resources. Please note the listings are not exhaustive, and inclusion of a course is not to be construed as an endorsement.
Refer to Section 5.4 and 5.4.1.1 of the NAPRA Guidance Document for Pharmacy Compounding of Non-sterile Preparations.
The College-adopted NAPRA Non-Sterile Compounding Standards do not specify a minimum size or dimensions for Level A compounding. There must be a separate space designated for compounding with an area large enough for compounding personnel to work comfortably and safely, with room to store equipment and products in an orderly manner in clean and secure surroundings. Also, the area should be designed and arranged to prevent cross-contamination between products, and it should be located away from parts of the pharmacy where there is a considerable amount of traffic (e.g., aisles, entrance and exit).
Refer to Sections 1, 2 the NAPRA Guidance Document for Pharmacy Compounding of Non-sterile Preparations.
The College adopted NAPRA Model Standards for Pharmacy Compounding of Non-Sterile Preparations and Guidance Document for Pharmacy Compounding of Non-sterile Preparations, as the standard for non-sterile compounding in Ontario.
Standards outline the minimum mandatory expectations that must be met by the profession, using the language of “must.” Therefore, these Model Non-Sterile Compounding Standards represent the minimum requirements to be applied in compounding to ensure the overall quality and safety of non-sterile preparations.
Each standard has a corresponding section in the Guidance Document with details concerning how the standard can be achieved. The Guidance Document was developed by NAPRA as a supplemental resource and establishes professionally-accepted means by which pharmacies can achieve compliance with the standards.
Where the guidance uses the language of “should”, it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested, if they have been proven to be equivalent or superior to those described in the Guidance Document.
Refer to Sections 3 and 5 of the NAPRA Guidance Document for Pharmacy Compounding of Non-sterile Preparations.
Compounding is within the scope of practice and authorized acts for pharmacy professionals defined in the Pharmacy Act, and compounding non-sterile preparations according to recognized guidelines and standards is an entry-to-practice competency for both pharmacists and pharmacy technicians. Therefore, it is reasonable that the public and other health practitioners expect any pharmacy to provide some compounding services.
The requirements for “all levels of compounding” (Section 5), corresponding to Level A, should be attainable for all pharmacies already engaged in compounding. Moreover, the College‘s Code of Ethics expects that “registrants make every reasonable effort to provide quality cost-effective pharmacy care and services to patients and society.”
As explained in Section 3 of NAPRA’s Guidance Document, “given that pharmacists and pharmacy technicians are expected to maintain competency in basic compounding skills, they are also expected to provide compounded preparations within their level of expertise and within the limitations of available and appropriate facilities and equipment.”
As for all prescriptions, it is expected that a pharmacist will assess the prescription for a non-sterile preparation to determine whether it is the most appropriate therapy for the particular patient or if there is a suitable, safer alternative. If a compounded preparation is the best option, the Designated Manager/department head must ensure that the pharmacy has the resources necessary to safely and accurately prepare a quality preparation. If not, they have an ethical obligation to “assume responsibility for making reasonable efforts to ensure continuity of patient care when they are unable to provide requested pharmacy services.”