Can you explain the NIOSH “hierarchy of controls” diagram depicting various levels of controls that can be implemented to mitigate risk?
The National Institute for Occupational Safety and Health (NIOSH) website explains that a hierarchy of controls is way to identify a preferred order of actions to best control hazardous workplace exposures. Compounding supervisors should review the descriptions of each level of control. The diagram below* provides examples of what the various controls might be feasible in the context of pharmacy compounding to minimize risk of exposure to occupational hazards.
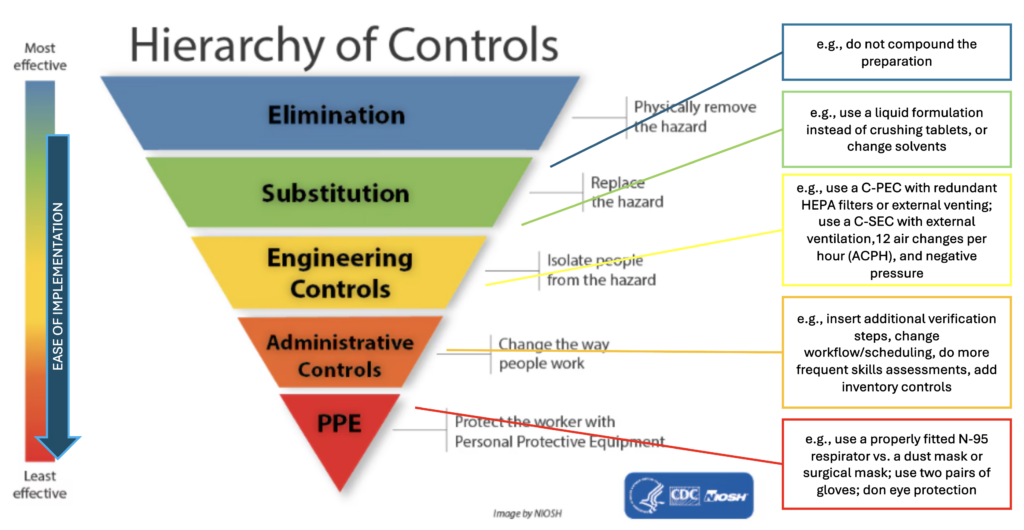
*Adapted from: https://www.cdc.gov/niosh/topics/hierarchy/default.html.
It is the responsibility of the compounding supervisor and manager to evaluate and choose the appropriate controls, depending on the type of risk posed by a hazardous product. Key points to consider in your decision-making are:
- The level of risk the ingredient(s) may present
- The volume/frequency of each ingredient used
- The combined exposure to all higher risk ingredients
Each prescription and patient situation must be assessed individually. In the event it is necessary to compound a particular preparation requiring processes and safety measures that are not currently in place in the pharmacy, the documentation should specify the following:
- Potential risks of compounding the preparation without the processes and safety measures currently in place
- The extra steps that must be taken to mitigate these risks
- References confirming that these steps will actually minimize risks to the quality of the product and safety of personnel
Procedures for mitigating risk must be documented in the master formulation record and reviewed at least every 12 months.
Refer to sections 9, 9.1.1, 9.2.1, 9.2.3 and 9.6 of the NAPRA Guidance Document for Pharmacy Compounding of Non-Sterile Preparations and the Pharmacy Connection article — A Closer Look at Personal Protective Equipment. Note that where the guidance document uses the language of “should,” it may be acceptable to meet the required standard using other technologies, techniques, materials and procedures than those suggested only if they have been proven to be equivalent or superior to those described in the guidance document.